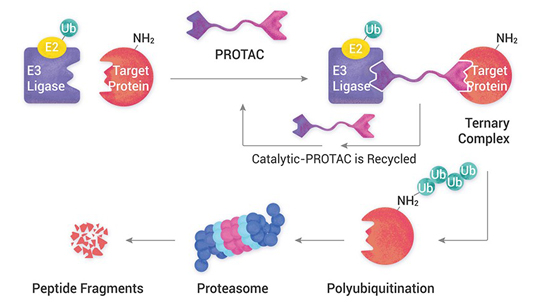
APSL has a legacy of developing 500+ molecules, several of which are in the commercial phase in highly regulated markets such as the USA, Europe, Japan, etc. We support our customers at various stages of a late-phase product from regulatory filing to launch and at the patent expiry stage with cost optimization options. Our vast manufacturing footprint allows us the flexibility to choose from a variety of unit operations suitable for product manufacturing. Our expertise in regulatory filings and documents will help our customers create comprehensive documentation for NDA and other global regulatory filings.
We offer a wide range of manufacturing services from a few kilos to multi tons of cGMP manufacturing of Key Starting Material (KSM) / Registered Starting Materials (RSM), advanced intermediates, and APIs. Our facilities can handle a diverse set of chemical reactions at various scales. Our facilities are inspected by global regulatory authorities like the US FDA, UK MHRA, TGA Australia, PMDA Japan, and other regulatory agencies. Our facilities can handle the manufacturing of niche technologies like peptides, High Potent Active Pharmaceutical Ingredients (HPAPIs), steroids, Methoxy Polyethylene Glycols (m-PEGs), and prostaglandins. Our state-of-the-art facilities are located across the globe in India, the UK, and Mexico.


CGMP manufacturing scale
Reactor size
- Kilo lab: 50 to 630 L
- Commercial: 500 to 10,000 L
Capacity of Production
- Kilo lab: 1 to 10 Kg
- Commercial: 15 to 500 Kg
MOC
- All glass set-up, Mild Steel Glass Lined (MSGL), stainless steel (SS316), and Hastelloy
Niche reactions
- Cryogenic reactor size (2,000 to 11,000 L) with Reaction temperature up to -80 deg C
- Hydrogenator capacity (20 to 5000 L) at 4 to 3535 bars
Downstream operations
Centrifuge
- 12”, 36” and 48”
Nustche Filters
- 25 L to 1 KL
Agitated Nustche Filter Dryer (ANFD)
- 0.1 m2 to 5.0 m2
Spray Dryers
- 5 to 50 Kg/Hr
Micronization
- Development (M50)
- Manufacturing (M100 to M2000
All manufacturing facilities are equipped with particle size regulating equipment including multi-mill, air jet mill or micronizer, sifter, and blenders. Dedicated unit for the micronization of steroid products. Steroid manufacturing facility is also equipped with micronizer.
Steroid
Development/PoC
- Quantity: 10 to 50 g
- Capacity: Lab scale
Pre-clinical/NGMP
- Quantity: 100 to 250 g
- Capacity: 20 to 50 L
Early Phase
- Quantity: 1 to 3 kg
- Capacity: 20 to 189 L
Late Phase
- Quantity: 5 to 10 Kg
- Capacity: Up to 7,000 L
Commercial
- Quantity: 25 to 100 Kg
- Capacity: Up to 7,000 L
HPAPI
Development/PoC/Non GMP
- Quantity: 10 to 50 g
- Capacity: Lab scale
Pre-clinica
- Quantity: 100 to 250 g
- Capacity: Lab scale
Early Phase
- Quantity: 1 to 3 kg
- Capacity: ~20 to 2,000 L
Late Phase
- Quantity: 5 to 10 Kg
- Capacity: 160 to 2,000 L
Commercial
- Quantity: 10 to 15 Kg
- Capacity: 160 to 2,000 L
PEGylation
Development/PoC
- Quantity: 10 to 50 g
- Capacity: Lab scale
Pre-clinical/NGMP
- Quantity: 250 to 500 g
- Capacity: Lab scale
Early Phase
- Quantity: 25 to 50 kg
- Capacity: 50 to 3,000 L
Late Phase
- Quantity: 100 to 250 Kg
- Capacity: 50 to 3,000 L
Commercial
- Quantity: 500 to 1,000 Kg
- Capacity: 50 to 3,000 L
Carbohydrate
Development/PoC
- Quantity: Up to 100 g/batch
- Capacity: Lab scale
Commercial
- Quantity: Up to ~30 Kg/batch
- Capacity: 60 to 250 L
Purification: GPC technique, size-exclusion chromatography, SAX purification, Ion-exchange chromatography
Analytical capability at the plant
Chromatography
- HPLC (UV, PDA, ELSD & RI detectors with Empower 3 software)
- GC (FID, TCD, ECD detectors with Empower 3 software)
- UPLC instruments
Characterization
- Mass spectrometer (LC-MS & HR-MS)
- AB SCIEX 4500 QQQ instrument
- Waters UPLC with LCT premier XE time of flight detector
- Agilent HPLC with 6410 triple quad mass detector
- Agilent GC with 5975C EI/CI Inert XL detector
Spectroscopy and Thermal analysis
- UV-Vis, IR, NMR spectroscopy, and photometers
- DSC and TGA
- Surface area analyzers
Sustainable manufacturing
Sustainability is one of our core values and we continue to build on our goals aligned with global standards. Our work in the field of sustainability was recognized by:
- DJSI (Dow Jones Sustainability Indices)
- FTSE4Good Index
- CDP (Carbon Disclosure Project)
- S&P BSE Carbonex
- S&P BSE Greenex
- Bloomberg Gender-Equality Index
- CII-SR EHS Excellence Silver Award
- PSCI Audited
Regulatory
Our development center is a US FDA audited facility, and our manufacturing plants are regularly audited by the US FDA, EDQM, KFDA (Korea), MHRA (UK), PMDA, SFDA, CDSCO, DMA, TGA, WHO GMP, and COFEPRIS.
Why Aurigene Intermediates and API Contract Manufacturing Services?
US FDA Inspected Labs
Integrated Development and Manufacturing
25 to 50 gms/batch
Dedicated Analytical Lab
ELN
Connect with our scientific experts for your drug discovery, development, and manufacturing needs
We understand that clear communication is essential to successful collaborations, and that's why we have a dedicated team that is always ready to help you. Whether you have questions about our services, want to discuss a potential partnership, or simply want to learn more about our company, we're here to help.
Our team of experts is dedicated to providing personalised solutions tailored to your unique needs. So, please don't hesitate to reach out to us. We look forward to hearing from you and helping you achieve your business goals.
Resources
FEBRUARY 25, 2025
Transforming Drug Discovery with Aurigene.AI
In the early 2000s, developing Sovaldi, a hepatitis C treatment, took over a decade and nearly $2 billion. Similarly, Zolgensma, a gene therapy for spinal muscular atrophy, required 15 years due to its complexity. However, the advent of artificial intelligence (AI) has revolutionized drug discovery. For example, in 2022, Pfizer's PAXLOVID, an oral COVID...
Read More
Advancement in personalized medicine and how the CRDMO industry is part of the solution
Personalized medicine is transforming the healthcare landscape by customizing treatment plans to individual patients’ unique genetic, clinical and environmental characteristics. These are effective and less invasive treatments for a wide range of conditions. Contract Research, Development and Manufacturing Organizations (CRDMOs) play an important role...
Read More
Cell Line Development
We enable development of stable and high yielding recombinant Mammalian and Microbial lines. ...
Read MoreFamiliarization, process optimization, and cGMP manufacturing and supply of 30.0 kg of a Bioactive Nucleotide (NAD Booster)
Background: A US-based biopharmaceutical company approached Aurigene Pharmaceutical Services for the familiarization, process development, and cGMP manufacturing and supply of 30.0 kg Nucleotide product (NAD booster) for phase-appropriate studies. The synthesis of the desired product involves three linear stages, which starts with reaction of a pentose...
Read MoreIdentification of Degradants of Thermal and Oxidation Stress Studies of Empagliflozin and Linagliptin Tablets by HPLC-PDA and LC-MS Instrumental Techniques
2022
Objective of the manuscript is to identify the degradants observed in the thermal and oxidation degradation sample of Empagliflozin and Linagliptin tablets by using LC-MS and HPLC-PDA instrumental techniques. Thermal and oxidation degradation samples were injected in HPLC-PDA and LC-MS instruments. Mass of the degradants were detected by LC-MS technique, ...
Read More-
Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action.
2005
Mutations in MEK1/2 have been described as a resistance mechanism to BRAF/MEK inhibitor treatment. We report the discovery of a novel ATP-competitive MEK1/2 inhibitor with efficacy in wildtype (WT) and mutant MEK12 models. Starting from a HTS hit, we obtained selective, cellularly active ...
Read More -
Wang-OSO3H catalyzed green synthesis of bioactive isoindolo[2,1- a ]quinazoline-5,11–dione derivatives: An unexpected observation
2005
The sulphonic acid-functionalized Wang resin (Wang-OSO3H) was explored as a polymeric and recov- erable acidic catalyst for the synthesis of isoindolo[2,1- a ]quinazoline-5,11–dione derivatives under green conditions. Thus the Wang-OSO3H ...
Read More -
Polycyclic Aromatic Compounds: A Simple and Efficient [(n-Bu3Sn)2MO4]n Catalyzed Synthesis of Quinazolinones and Dihydroquinazolinones
2005
A novel unprecedented approach for the synthesis of various quinazolinones and dihydroquinazolinones has been using [(n-Bu3Sn)2MO4]n as a catalyst. The reaction has been screened ...
Read More
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market