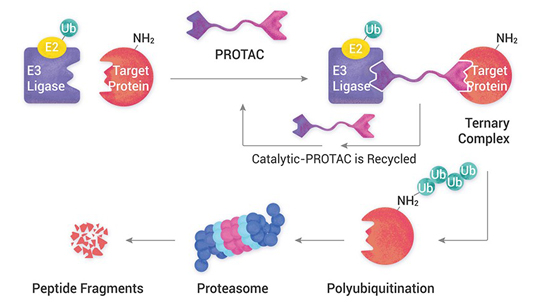
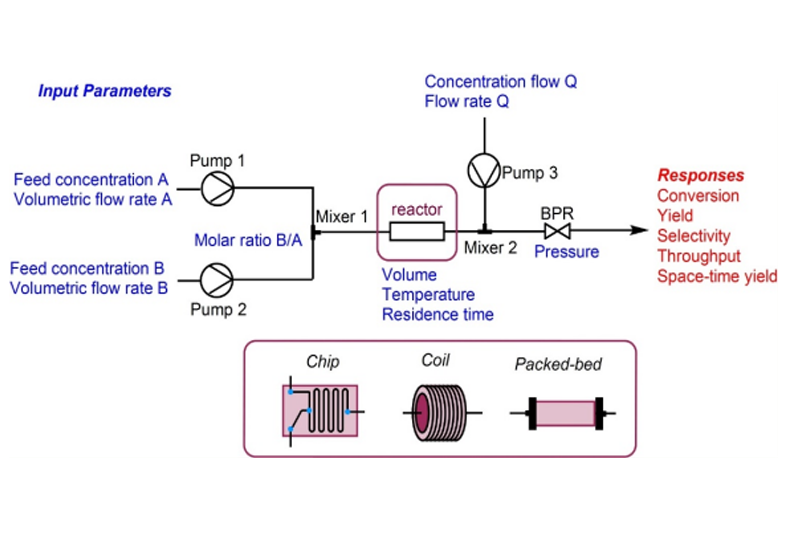
Our dedicated team of highly skilled scientists from various disciplines conducts comprehensive toxicology studies. We perform GLP-compliant analytical and bioanalytical method validations, formulation analysis and bioanalysis to support preclinical toxicology studies. These validations and analyses are carried out for plasma drug level determinations and active ingredient content, homogeneity and stability of the formulations used in non-clinical safety studies.
Our vivarium, accredited by AAALAC, features state-of-the-art laboratories where clinical pathology analyses and histopathology procedures are conducted. We perform rodent toxicity studies in rats and mice, including repeat dose toxicity studies over a defined period of test article administration, with or without a recovery period.
The facility is GLP certified by the National GLP Monitoring Authority (NGCMA) of India. We offer repeat dose 4-day, 7-day, or 14-day dose range DRF toxicity studies in rats and mice. These studies aim to identify toxic effects over specific exposure periods and determine the Maximum Tolerated Dose (MTD) and/or No-Observed-Adverse-Effect-Level (NOAEL). The findings from DRF studies helps in the selection of dosage regimens for subsequent long-term or definitive GLP toxicology studies in rats and mice.



Exploratory Toxicology Studies
We offer a comprehensive range of exploratory toxicology studies, including single dose toxicity studies and 4-day, 7-day, or 14-day repeat dose toxicity studies. A single dose escalation phase is often integrated with the DRF study to gather maximum data. These studies aim to determine the Maximum Tolerated Dose (MTD) and/or No-Observed-Adverse-Effect-Level (NOAEL) over a short period of exposure through the intended clinical route in humans.
Our study designs are tailored to meet the client requirements considering the nature of the test compounds and intended product development program. Typical end points include mortality checks, clinical signs observations, body weight and feed intake measurements, clinical pathology, gross pathology observations, and limited histopathology.
GLP Definitive Toxicology Studies
We conduct GLP toxicology studies in rats and mice to support IND and ANDA submissions. These studies include acute, subacute (ranging from 7 to 28 days), subchronic (90 days), chronic (180 days to 1 years) toxicity and carcinogenicity studies. Studies are conducted as per the relevant regulatory guidelines (FDA, OECD, ICH, MHRA etc.) in AAALAC accredited animal facilities.
The end points of these toxicity studies include mortality checks, clinical signs of toxicity, body weight and food consumption measurements, interim/terminal clinical pathology (including haematology, clinical pathology and urinalysis), gross pathology and histopathology. Our highly experienced toxicologists conduct these studies in AAALAC-accredited facilities. Our clinical pathology and histopathology labs are equipped with GLP-compliant instruments from internationally reputed suppliers. The computerized systems used in our laboratories are validated and fit for purpose as required by the Principles of GLP based on the study design, satellite toxicokinetic (TK) groups are added, if required, to obtain plasma drug exposure at scheduled intervals (e.g., first and last days of dosing). To support formulation analysis and bioanalysis, our Toxicology laboratory coordinates with GLP compliant Analytical and DMPK facilities.
4-Day/7-Day/14-Day Repeat Dose DRF Toxicology Studies
The end points of the DRF toxicity studies include mortality checks, observation of clinical signs of toxicity, body weight and food consumption measurements, terminal clinical pathology (hematology, limited or comprehensive clinical pathology, urinalysis etc.), gross pathology and limited or comprehensive histopathology. As required by the study design, satellite toxicokinetic (TK) groups are added to obtain plasma drug exposure at scheduled intervals (e.g., first and last days of dosing). Based on client requirement, analysis of dose formulation samples will be included to estimate the drug concentration verification and homogeneity analysis. To support formulation analysis and bioanalysis, the Toxicology laboratory coordinates with GLP compliant Analytical and DMPK facilities located within the premises.
The in-life phase of these studies are performed by a group of highly experienced toxicologists in AAALAC accredited animal facilities. Our clinical pathology and the histopathology labs are state-of-the-art facilities equipped with GLP compliant instruments from internationally reputed suppliers. The computerized systems used in these laboratories are validated and fit for purpose as required by the principles of GLP.
Maximum Tolerated Dose (MTD) Studies
The main objectives of the single dose Maximum Tolerated Dose (MTD) studies is to determine the maximum dose level that is tolerated by the test species and to determine the dose levels for the subsequent repeat dose toxicity studies by the intended clinical / therapeutic exposure. In the absence of a relevant literature/data for a specific highest test dose, we test up to 2000 mg/kg as a limit dose in line with the recommendations of various international guidelines for conducting acute toxicity studies.
The study is conducted by assigning rats or mice into different treatment groups and exposing them to various graded dose levels to ascertain the toxicity at each dose level and determine the MTD. Essentially, the study end points cover mortality checks (at least twice daily), clinical signs examination for identification of evident toxicity (routine/detailed), body weight and food consumption measurements, gross pathology and as required by the study protocol and a limited histopathology.
We also conduct extended single-dose toxicity studies as per ICH guidelines that include, clinical pathology (hematology, clinical biochemistry, & comprehensive histopathology) in addition to the above stated study end points. Formulation analysis for the GLP toxicity studies are performed as required by the client.
Subacute Toxicity Studies
Subacute toxicity studies are typically designed as per guidelines such as FDA, OECD, ICH, DCGI and MHRA to evaluate the adverse effects of a substance when administered repeatedly over a short period, typically ranging from 14 to 28 days. These studies are crucial for understanding the potential toxic effects of a substance before it progresses to longer-term studies or clinical trials.
The primary objectives of subacute toxicity studies include:
- Identifying target organs: Determining which organs or systems are most affected by the substance.
- Establishing dose-response relationships: Understanding how different doses of the substance affect the severity and type of toxic effects.
- Determining NOAEL and LOAEL: Identifying the No-Observed-Adverse-Effect Level (NOAEL) and the Lowest-Observed-Adverse-Effect Level (LOAEL).
Subacute toxicity studies in rats and mice typically involve:
- Dosing: Animals are administered the test substance daily, starting at a dose expected to be near the therapeutic level and increasing stepwise until toxic signs are observed
- Duration: The study duration is usually between 14 and 28 days.
During the study, various parameters are monitored to assess the toxic effects:
- Clinical Observations: Regular checks for signs of toxicity, such as changes in behavior, appearance, and physiological functions.
- Body Weight and Food Consumption: Monitoring changes in body weight and food intake to detect any adverse effects on growth and metabolism.
- Clinical Pathology: Blood and urine tests to evaluate the impact on hematology, clinical chemistry, and urinalysis.
- Gross and Histopathology: Examination of organs and tissues at the end of the study to identify any pathological changes.
Subacute toxicity studies play a vital role in the early stages of drug development and chemical safety evaluation, helping to identify potential risks and inform the design of longer-term studies.
Why Aurigene Pharmaceutical Services
Expertise in designing and conducting studies to fulfil requirements of FDA, OECD, ICH, MHRA regulatory guidelines
Highly experienced and expert toxicology and analytical scientists
AAALAC-accredited animal facility
State-of-the-art facility equipped with GLP compliant instruments
Wide range of toxicology services
Connect with our scientific experts for your drug discovery, development, and manufacturing needs
We understand that clear communication is essential to successful collaborations, and that's why we have a dedicated team that is always ready to help you. Whether you have questions about our services, want to discuss a potential partnership, or simply want to learn more about our company, we're here to help.
Our team of experts is dedicated to providing personalised solutions tailored to your unique needs. So, please don't hesitate to reach out to us. We look forward to hearing from you and helping you achieve your business goals.
Resources
FEBRUARY 25, 2025
Transforming Drug Discovery with Aurigene.AI
In the early 2000s, developing Sovaldi, a hepatitis C treatment, took over a decade and nearly $2 billion. Similarly, Zolgensma, a gene therapy for spinal muscular atrophy, required 15 years due to its complexity. However, the advent of artificial intelligence (AI) has revolutionized drug discovery. For example, in 2022, Pfizer's PAXLOVID, an oral COVID...
Read More
Advancement in personalized medicine and how the CRDMO industry is part of the solution
Personalized medicine is transforming the healthcare landscape by customizing treatment plans to individual patients’ unique genetic, clinical and environmental characteristics. These are effective and less invasive treatments for a wide range of conditions. Contract Research, Development and Manufacturing Organizations (CRDMOs) play an important role...
Read More
Cell Line Development
We enable development of stable and high yielding recombinant Mammalian and Microbial lines. ...
Read MoreFamiliarization, process optimization, and cGMP manufacturing and supply of 30.0 kg of a Bioactive Nucleotide (NAD Booster)
Background: A US-based biopharmaceutical company approached Aurigene Pharmaceutical Services for the familiarization, process development, and cGMP manufacturing and supply of 30.0 kg Nucleotide product (NAD booster) for phase-appropriate studies. The synthesis of the desired product involves three linear stages, which starts with reaction of a pentose...
Read MoreIdentification of Degradants of Thermal and Oxidation Stress Studies of Empagliflozin and Linagliptin Tablets by HPLC-PDA and LC-MS Instrumental Techniques
2022
Objective of the manuscript is to identify the degradants observed in the thermal and oxidation degradation sample of Empagliflozin and Linagliptin tablets by using LC-MS and HPLC-PDA instrumental techniques. Thermal and oxidation degradation samples were injected in HPLC-PDA and LC-MS instruments. Mass of the degradants were detected by LC-MS technique, ...
Read More-
Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action.
2005
Mutations in MEK1/2 have been described as a resistance mechanism to BRAF/MEK inhibitor treatment. We report the discovery of a novel ATP-competitive MEK1/2 inhibitor with efficacy in wildtype (WT) and mutant MEK12 models. Starting from a HTS hit, we obtained selective, cellularly active ...
Read More -
Wang-OSO3H catalyzed green synthesis of bioactive isoindolo[2,1- a ]quinazoline-5,11–dione derivatives: An unexpected observation
2005
The sulphonic acid-functionalized Wang resin (Wang-OSO3H) was explored as a polymeric and recov- erable acidic catalyst for the synthesis of isoindolo[2,1- a ]quinazoline-5,11–dione derivatives under green conditions. Thus the Wang-OSO3H ...
Read More -
Polycyclic Aromatic Compounds: A Simple and Efficient [(n-Bu3Sn)2MO4]n Catalyzed Synthesis of Quinazolinones and Dihydroquinazolinones
2005
A novel unprecedented approach for the synthesis of various quinazolinones and dihydroquinazolinones has been using [(n-Bu3Sn)2MO4]n as a catalyst. The reaction has been screened ...
Read More
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market