
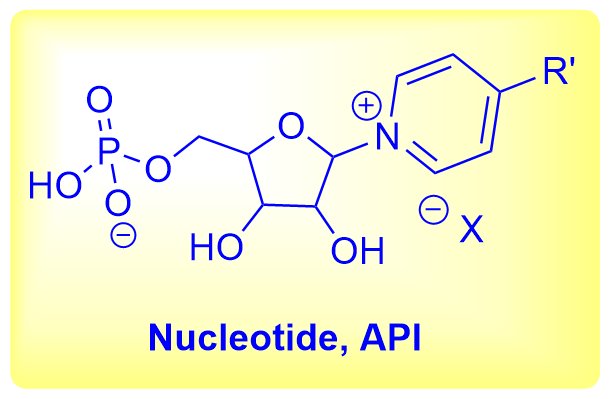
Background: A US-based biopharmaceutical company approached Aurigene Pharmaceutical Services for the familiarization, process development, and cGMP manufacturing and supply of 30.0 kg Nucleotide product (NAD booster) for phase-appropriate studies.
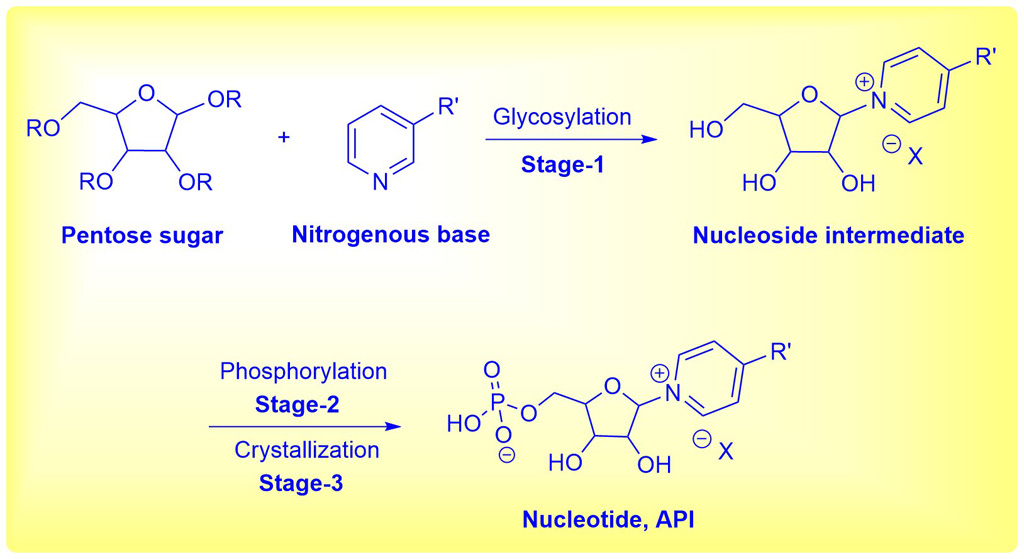
The synthesis of the desired product involves three linear stages, which starts with reaction of a pentose sugar derivative with a nitrogenous base to give a nucleoside intermediate, which on phosphorylation yields the crude nucleotide intermediate. In the final stage, the crude intermediate was purified using a special grade silica gel to afford the pure nucleotide product.
Challenges: Stage-1 reaction mass has limited stability with low product conversion in the optimized reaction solvent. Handling of Stage-1 and Stage-2 intermediates is challenging owing to their highly hygroscopic nature. The Stage-2 reaction has to be maintained at -20 ºC for approximately 72 hours to achieve the desired conversion. Phosphorylation reaction at higher temperatures (i.e., above -15 ºC) would result in undesired impurities with poor conversion. Identifying a suitable purification protocol for all the stages to achieve the desired quality is challenging. Removal of polar impurities in API purification process is a key challenge. Scaling of product in the reaction vessel during API crystallization process poses operational and yield challenges. A major challenge associated with the API stage is meeting the specification of eight known impurities and any individual unspecified impurity in RS by HPLC analysis. Aurigene solution: Optimization of all the processes to develop scale-friendly methods while establishing well-defined process controls. Identified that presence of non-volatile impurities in reaction solvent results in low product conversion. Quickly fixed the issue by using distilled solvent to achieve high conversion, >97% purity and quantitative yield in Stage-1. Developed QTLC (Quantitative Thin Layer Chromatography) for quick analysis to assess the reaction conversion in Stage-1. Optimized the stoichiometry of phosphorous oxychloride in Stage-2. Scale the suitable isolation process developed in Stage-2. Crystallization studies performed to identify the seeding point to avoid scaling in the reactor. An optimized crystallization process was developed in the API stage to meet the specifications of all impurities and achieve constant yield & quality. Special grade silica gel employed for the removal of polar impurities from crude nucleotide intermediate in API stage. This silica gel is recovered, recycled and reused for over 10 times in the lab on a gram scale and over 2 times in scale up batches on a 100 kg scale. Proactively mapped and commissioned TCU (Temperature control unit) for uninterrupted maintenance of -20 ºC in reactor and ANFD (Agitated Nutsche Filter Dryer) to filter the highly hygroscopic intermediates in scale up batches. Outcome:
Optimized processes performed well at scale, affording consistent yield and quality, and enabled smooth execution. TCU helped to attain -20 oC in Stage-2 scale up batches without any challenges. Similarly, ANFD aided in hassle-free filtration of hygroscopic intermediates in manufacturing batches. Completely avoided product scaling in the reactor by adopting the developed crystallization process in the scale-up batches of API. Manufactured 30.0 kg of API and delivered to customer. Green chemistry principles adopted by recovering, recycling, and reusing the special grade silica gel. Nucleotide platform established at Aurigene with this project.
Conclusions: Typically, this project requires appropriate engineering controls for scale up batches such as (i) TCU to maintain cryogenic reaction, (ii) ANFD to handle hygroscopic intermediates, (iii) Customized PNF (Pressure Nutsche Filter) in final stage for purification of crude API using a special grade silica gel, etc., along with holistic understanding of all the stages. Aurigene was proactive in identifying the required engineering controls, commissioned desired equipment, successfully demonstrated the scale up batches, right first time and delivered the product to customer. Aurigene team received customer appreciation for lab development work, establishing suitable in-process controls and smooth scale up execution. Overall, Aurigene showcased its inherent expertise, capability, skill, competency and manufacturing ability to handle new technologies from this case study.
Contact Us
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market
