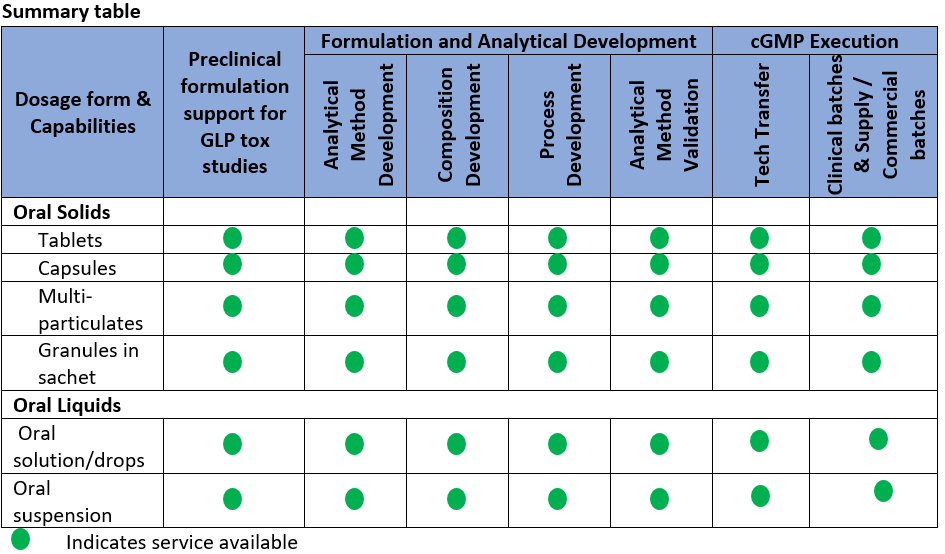
Starting with comprehensive pre-formulation services to commercial manufacturing, we provide a full suite of services for formulations of oral, parenteral and topical dosage forms. We offer “Fit-for-purpose” formulation development for pre-clinical and First in Human (FIH) studies based on molecule’s physico-chemical behavior, excipient compatibility studies, and quick screening of excipients.
Our state- of-the-art formulation manufacturing facilities are in Hyderabad and Visakhapatnam and are audited by all major regulatory agencies.
Cross-functional teams such as DMPK, API synthesis, salt screening, toxicology, process engineering, analytical development and regulatory teams are located in the same campus and work in an integrated manner at each stage of the life cycle of the molecule for efficient and on-time delivery.






Service Offerings:
Oral Dosage Forms
We offer integrated oral dosage form services right from pre-formulation services to commercial manufacturing. We provide recommendations for best dosage form to clinic, based on the preformulation study results and help customer to choose the phase appropriate dosage form that suites the therapeutic objective.
Pre-formulation services:
Our pre-formulation services include:- API physiochemical characterization
- Molecule stability behavior
- Solubility enhancement by various techniques
Based on API characteristics we provide recommendations for maximizing the bioavailability of the molecule through solubility and permeability enhancement, gastro retentive formulation and targeted drug delivery systems.
We offer pre-clinical supplies of formulations for animal studies from R&D. Our state of art lab facilities are equipped with miniature lab equipment that enables fit for purpose formulation development using minimum batch size as low as 50 g blend using minimal drug substance quantities.
We also offer support to veterinary formulations for development of customized formulation.
“Phase appropriate” dosage form development services for early phase clinical trials (Phase 1&2):
Insights from the pre-formulation and preclinical studies would be taken into consideration while designing dosage forms like tablets, capsules, oral liquids.
In addition to conventional approaches, we have expertise in
- Masking the taste of the bitter tasting drugs, support to select the type and level of sweetener, flavours, complexation agents using ‘e-tongue technology’.
- Xcelodose technology to directly fill the API in capsule with an accuracy of about 100 microgram to bring your NCE molecule right from preclinical stage to clinical stage in a cost effective manner.
- Multi unit particulate systems (MUPS) to achieve tailored drug release of actives in fixed dose combination to enable immediate and modified release of multiple active ingredients.
Late phase formulation development for Phase 3/Commercial supply:
We offer wide variety of dosage forms for late phase development up to commercial manufacturing. Our state of art laboratory equipment help to develop robust formulation using QbD approach and evaluate impact of formulation variables and critical material/process variables on drug product quality attributes during the lab scale development and provide recommendations for successful scale up and registration batches.
In addition to traditional dosage forms, we have the capability to develop and manufacture highly complex formulations involving bi-layer tablets, multi-particulate systems (Pellets or Mini-tablets in capsules), fixed dose combinations, very low and high-dose products, modified release formulations, oral solutions and suspensions. We provide patient centric solutions for the dosage form design.
We have access to state of art manufacturing facilities that are approved by US FDA, MHRA and WHO to enable seamless cGMP manufacturing of clinical/registration and commercial batches. Most of the manufacturing facilities follow paperless electronic BMR systems and comply to stringent regulatory requirements.
Parenteral Dosage Forms
Our depth and breadth of experience and specialization in parenteral dosage form development is remarkable which can provide insight and innovation. We offer the expertise in overcoming the challenges associated with parenteral drug product development such as but not limited to.
- Solubility enhancement studies
- Achieving a good compatibility of the drug substances with the excipients
- Establishing the compatibility of the preparations with the container closure system
- Stability challenges
A constant focus on key control measures from pre-formulation development through drug product development to GMP manufacturing is provided to meet time-to-market schedules within the prescribed budget. With a portfolio of options to choose from, we partner with customers to find the approach that best achieves their technical goals.
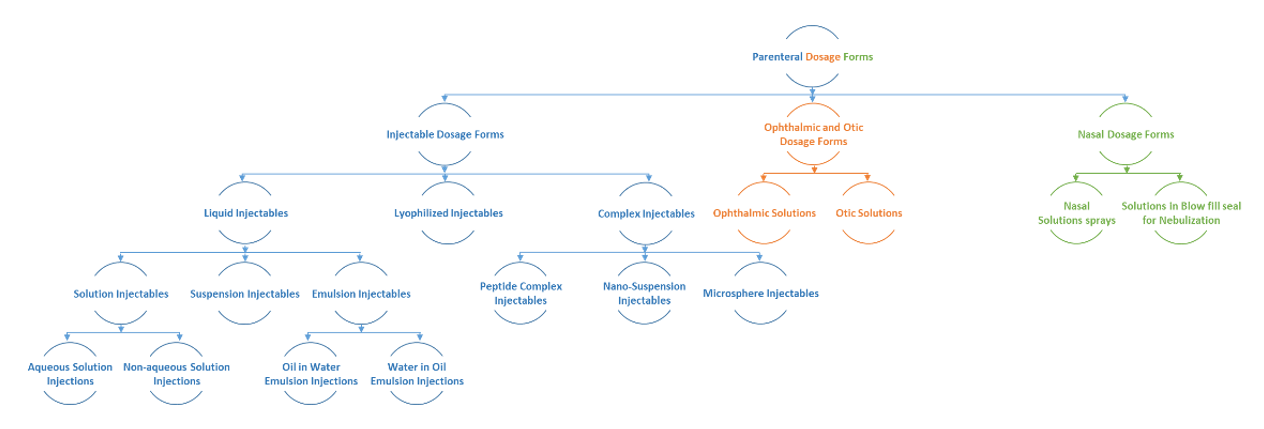
Fig: Flow diagram for services offered in Parenteral dosage forms
Parenteral formulation development includes following activities but not limited to identification, selection and optimization of:
- Formulation composition (solubility enhancers, isotonicity agents, chelating agents, bulking agents, anti-oxidants, preservatives etc)
- Process and process parameters (mixing speed and mixing time, process temperature, order of addition of excipients etc)
- Product Container closure system
- Other supporting studies such as in-use stability studies, preservative effectiveness testing, compatibility and hold time studies and stability studies, reconstitution studies and dilution studies
Our parenteral formulation development lab includes all the required equipments and also critical equipment such as
- Martin Christ 2-10D LSC plus pilot scale lyophilizer
- Bead Mill
- High pressure homogenizer
- Fedegari counter pressure autoclave
Our State of art manufacturing facilities for parenteral dosage forms are located at vishakapatnam and are approved by major regulatory agencies (MFDS/USFDA/MHRA)
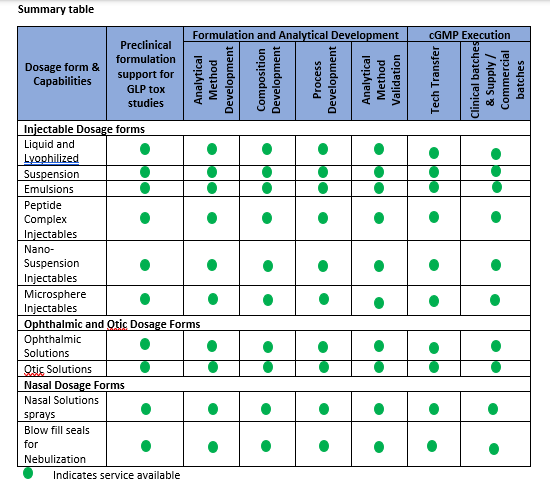
Parenteral Dosage Forms
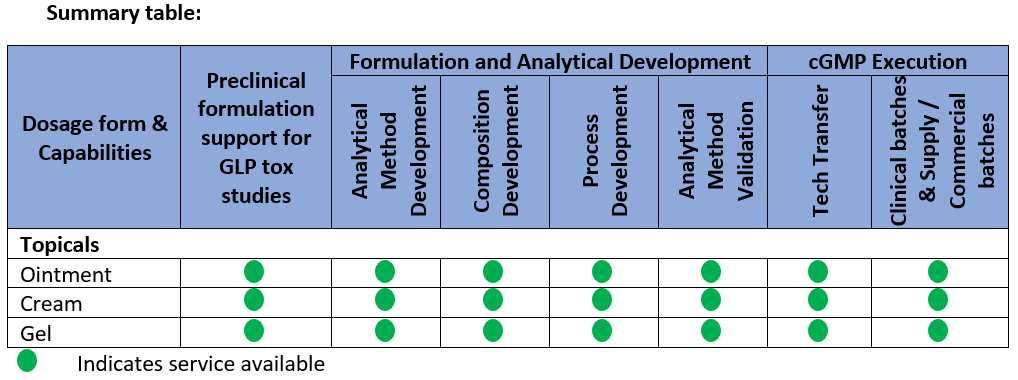
We offer formulation development through a skilled scientific team with expertise in Topical dosage forms both in conventional and microspheres based formulations
- Ointments
- Creams
- Gels
We provide tailor made solutions to address the challenges of topical dosage forms and help to optimize the development time and costs
Why Aurigene Pharmaceutical Services?
Fit-for-purpose development strategy
Integrated cross-functional teams such as PK-PD, API synthesis, salt screening, toxicology, process engineering, and analytical development teams
Services across the product lifecycle
USFDA inspected GMP facilities
Connect with our scientific experts for your drug discovery, development, and manufacturing needs
We understand that clear communication is essential to successful collaborations, and that's why we have a dedicated team that is always ready to help you. Whether you have questions about our services, want to discuss a potential partnership, or simply want to learn more about our company, we're here to help.
Our team of experts is dedicated to providing personalised solutions tailored to your unique needs. So, please don't hesitate to reach out to us. We look forward to hearing from you and helping you achieve your business goals.
Resources
FEBRUARY 25, 2025
Transforming Drug Discovery with Aurigene.AI
In the early 2000s, developing Sovaldi, a hepatitis C treatment, took over a decade and nearly $2 billion. Similarly, Zolgensma, a gene therapy for spinal muscular atrophy, required 15 years due to its complexity. However, the advent of artificial intelligence (AI) has revolutionized drug discovery. For example, in 2022, Pfizer's PAXLOVID, an oral COVID...
Read More
Advancement in personalized medicine and how the CRDMO industry is part of the solution
Personalized medicine is transforming the healthcare landscape by customizing treatment plans to individual patients’ unique genetic, clinical and environmental characteristics. These are effective and less invasive treatments for a wide range of conditions. Contract Research, Development and Manufacturing Organizations (CRDMOs) play an important role...
Read More
Cell Line Development
We enable development of stable and high yielding recombinant Mammalian and Microbial lines. ...
Read MoreFamiliarization, process optimization, and cGMP manufacturing and supply of 30.0 kg of a Bioactive Nucleotide (NAD Booster)
Background: A US-based biopharmaceutical company approached Aurigene Pharmaceutical Services for the familiarization, process development, and cGMP manufacturing and supply of 30.0 kg Nucleotide product (NAD booster) for phase-appropriate studies. The synthesis of the desired product involves three linear stages, which starts with reaction of a pentose...
Read MoreIdentification of Degradants of Thermal and Oxidation Stress Studies of Empagliflozin and Linagliptin Tablets by HPLC-PDA and LC-MS Instrumental Techniques
2022
Objective of the manuscript is to identify the degradants observed in the thermal and oxidation degradation sample of Empagliflozin and Linagliptin tablets by using LC-MS and HPLC-PDA instrumental techniques. Thermal and oxidation degradation samples were injected in HPLC-PDA and LC-MS instruments. Mass of the degradants were detected by LC-MS technique, ...
Read More-
Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action.
2005
Mutations in MEK1/2 have been described as a resistance mechanism to BRAF/MEK inhibitor treatment. We report the discovery of a novel ATP-competitive MEK1/2 inhibitor with efficacy in wildtype (WT) and mutant MEK12 models. Starting from a HTS hit, we obtained selective, cellularly active ...
Read More -
Wang-OSO3H catalyzed green synthesis of bioactive isoindolo[2,1- a ]quinazoline-5,11–dione derivatives: An unexpected observation
2005
The sulphonic acid-functionalized Wang resin (Wang-OSO3H) was explored as a polymeric and recov- erable acidic catalyst for the synthesis of isoindolo[2,1- a ]quinazoline-5,11–dione derivatives under green conditions. Thus the Wang-OSO3H ...
Read More -
Polycyclic Aromatic Compounds: A Simple and Efficient [(n-Bu3Sn)2MO4]n Catalyzed Synthesis of Quinazolinones and Dihydroquinazolinones
2005
A novel unprecedented approach for the synthesis of various quinazolinones and dihydroquinazolinones has been using [(n-Bu3Sn)2MO4]n as a catalyst. The reaction has been screened ...
Read More
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market