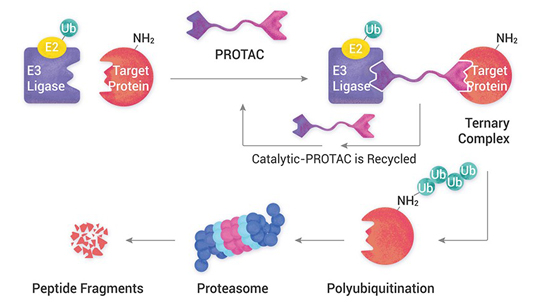
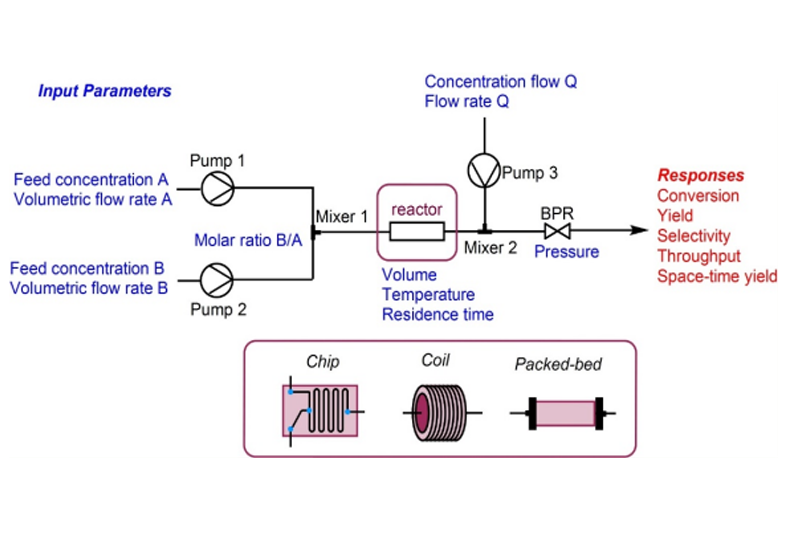
Analytical services are used to identify, isolate, or quantify molecules and to characterize their physical properties. We provide physiochemical and biological characterization services.
Backed by our strong protein chemistry background, we enable ‘Finger-Print’ protein structure and functional characterization for proteins from naked proteins to hyper-glycosylated or derivatized proteins.
Our biological characterization capability enables a sound understanding of the biological mechanism of action of the product. We can support a wide variety of biological assays including, cell-proliferation/killing, binding affinity, cell signaling, and viability or endpoint assays using both transformed cell lines and primary cells.




Physiochemical characterization
We provide physiochemical characterization services ranging from protein structure confirmation to amino acid content detection. We also quantify free cysteines and detect product impurities such as glycation and deamidation.
The ability is based on a diverse platform of instruments and technologies supported by a fully-trained technical staff:
- 21 CFR part 11 and GAMP compliant system-based labs
- A comprehensive instrumentation infrastructure to support orthogonal confirmation of product parameters
- Experience across diverse protein expression systems
Catalog:
- Primary structure: Intact mass, fragmented mass, and Pep map
- Higher order structure: Secondary and tertiary structure, melting point, disulfide linkage, and free thiol
- Post-translational modification: Oligosaccharide profiling, oxidation, deamidation, and cleavage
- Product/process - related impurities: Size and charge variant analysis
- Technology: Mass Spectrometry, HPLC, UPLC, spectroscopy, circular dichroism, fluorometry, DSC DLS, SEC MALS, ion chromatography, CEIEF, CESDS, and Solo VpE
- Protein: Mab, Fab, fusion protein, cytokine, enzyme, hormone, hyperglycosylated proteins, and derivatized proteins
Biological characterization
Our capability in luminescence and fluorescence-based assays and flow cytometry enables a wider reach of biological characterization. In addition, we also develop bio-assays for clinical sample analysis and immunogenicity assessment:
- Development of bioassays and cell-based assay employing primary and derived cell lines
- Expertise in luminescence, fluorescence, and flow cytometry-based assay
- Wide variety of cell signaling and endpoint assays – ADCC, CDC, and proliferation
- Ability to handle clinical samples
Catalog:
- Signaling: GPCR, calcium release, kinase, and phosphorylation
- Binding: Direct/indirect ELISA, competition ELISA, and surface plasmon resonance (Biacore)
- Viability: ADCC, CDC, and proliferation
- PK/PD: Serum drug concentration, immunogenicity, and neutralizing antibody
- Technology: Luminescence and fluorescence-based methods, multicolor flow cytometry, microscopy, and primary cell culture
- Protein: Mab, Fab, fusion protein, cytokine, enzyme, hormone, hyperglycosylated proteins, and derivatized proteins
Why Aurigene Analytical Services?
ALCOA+ driven quality systems
Diverse platform of instruments and technologies
Integrated approach to safety and quality management systems
In-house capability across the value chain with a trained scientific staff
Connect with our scientific experts for your drug discovery, development, and manufacturing needs
We understand that clear communication is essential to successful collaborations, and that's why we have a dedicated team that is always ready to help you. Whether you have questions about our services, want to discuss a potential partnership, or simply want to learn more about our company, we're here to help.
Our team of experts is dedicated to providing personalised solutions tailored to your unique needs. So, please don't hesitate to reach out to us. We look forward to hearing from you and helping you achieve your business goals.
Resources
FEBRUARY 25, 2025
Transforming Drug Discovery with Aurigene.AI
In the early 2000s, developing Sovaldi, a hepatitis C treatment, took over a decade and nearly $2 billion. Similarly, Zolgensma, a gene therapy for spinal muscular atrophy, required 15 years due to its complexity. However, the advent of artificial intelligence (AI) has revolutionized drug discovery. For example, in 2022, Pfizer's PAXLOVID, an oral COVID...
Read More
Advancement in personalized medicine and how the CRDMO industry is part of the solution
Personalized medicine is transforming the healthcare landscape by customizing treatment plans to individual patients’ unique genetic, clinical and environmental characteristics. These are effective and less invasive treatments for a wide range of conditions. Contract Research, Development and Manufacturing Organizations (CRDMOs) play an important role...
Read More
Cell Line Development
We enable development of stable and high yielding recombinant Mammalian and Microbial lines. ...
Read MoreFamiliarization, process optimization, and cGMP manufacturing and supply of 30.0 kg of a Bioactive Nucleotide (NAD Booster)
Background: A US-based biopharmaceutical company approached Aurigene Pharmaceutical Services for the familiarization, process development, and cGMP manufacturing and supply of 30.0 kg Nucleotide product (NAD booster) for phase-appropriate studies. The synthesis of the desired product involves three linear stages, which starts with reaction of a pentose...
Read MoreIdentification of Degradants of Thermal and Oxidation Stress Studies of Empagliflozin and Linagliptin Tablets by HPLC-PDA and LC-MS Instrumental Techniques
2022
Objective of the manuscript is to identify the degradants observed in the thermal and oxidation degradation sample of Empagliflozin and Linagliptin tablets by using LC-MS and HPLC-PDA instrumental techniques. Thermal and oxidation degradation samples were injected in HPLC-PDA and LC-MS instruments. Mass of the degradants were detected by LC-MS technique, ...
Read More-
Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action.
2005
Mutations in MEK1/2 have been described as a resistance mechanism to BRAF/MEK inhibitor treatment. We report the discovery of a novel ATP-competitive MEK1/2 inhibitor with efficacy in wildtype (WT) and mutant MEK12 models. Starting from a HTS hit, we obtained selective, cellularly active ...
Read More -
Wang-OSO3H catalyzed green synthesis of bioactive isoindolo[2,1- a ]quinazoline-5,11–dione derivatives: An unexpected observation
2005
The sulphonic acid-functionalized Wang resin (Wang-OSO3H) was explored as a polymeric and recov- erable acidic catalyst for the synthesis of isoindolo[2,1- a ]quinazoline-5,11–dione derivatives under green conditions. Thus the Wang-OSO3H ...
Read More -
Polycyclic Aromatic Compounds: A Simple and Efficient [(n-Bu3Sn)2MO4]n Catalyzed Synthesis of Quinazolinones and Dihydroquinazolinones
2005
A novel unprecedented approach for the synthesis of various quinazolinones and dihydroquinazolinones has been using [(n-Bu3Sn)2MO4]n as a catalyst. The reaction has been screened ...
Read More
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market