

We offer formulation development through a highly skilled scientific team with expertise in variety of dosage forms. At our state-of-the-art facilities, we handle simple and complex oral solid dosage forms which include tablets, capsules, powder for suspension and multi-particulate drug delivery systems customized for immediate release, modified release and targeted release.
We provide ‘Fit-for-Purpose’ formulation development to support early phase/ ‘First-in-Human’ studies and also provide end-to-end services up to commercial manufacturing. We also support for development of customized formulations for veterinary use and nutraceutical applications.
Our expert team is having experience in developing complex formulations and provide tailored solutions to your problems and help to optimize the development time and costs.






Service Offerings:
Solid Oral Dosage Forms
We offer extensive end-to-end services of formulation development for a wide variety of oral dosage forms. Our team assists from Prototype formulation screening studies, Formulation/process optimization using QBD principles, ICH compliant Stability studies, Scale-up/technology transfer, formulation development to development of First-in-Human (FIH) formulations to enable clinical trial studies from phase I to phase III/ commercial manufacture. We are equipped with state-of-the-art facilities to manufacture 500 to 50,000 units at R&D and up to 7,500,000 units at commercial manufacturing sites and complete CMC support for regulatory submission.
Technologies Offered
- Direct tablet compression
- Fluid bed processing (granulation, drying, coating)
- High-shear wet granulation and drying
- Dry granulation (roller compaction, slugging)
- Extrusion-Spheronization
- High-speed automated tablet compression
- Perforated pan tablet coating (mini tablets coating)
- Automated capsule filling (powder, granules, pellets, mini tablets)
- Low humidity/moisture requirements
- Light-sensitive compound handling
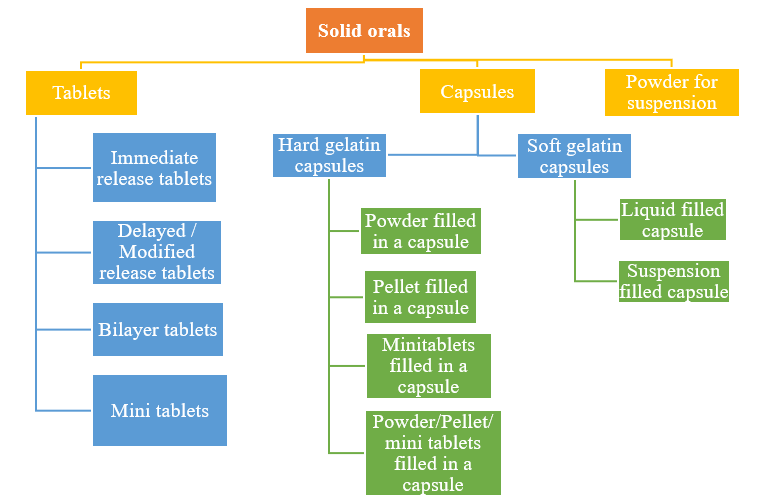
Tablets
Immediate Release, Delayed Release, Sustained Release, Oral Disintegrating, Film coated, Taste masking, Fixed Dose Combinations (FDC).
a. Single Layer Tablets
Depending on the mode of usage conventional uncoated or film coated immediate release tablets, orally disintegrating tablets, sublingual tablets, buccal tablets, taste masked tablets, effervescent tablets and chewable tablets can be developed
b. Delayed release/ modified release tablets
Proven expertise in formulation of acid labile drugs using delayed release technology and where necessary the drug release can be modulated or targeted to release at specific regions of the gastro-intestinal tract.
c. Bilayer Tablets
A combination of immediate release (loading dose) and modified release portions of a single drug can be incorporated in a single dosage form. Also, a combination of two or more drugs which may be incompatible can be formulated based on the requirement
d. Mini Tablets Fixed dose combination (FDC) or standalone drug molecules can be formulated as minitablets whi>ch offer the advantage of being multi-unit particulate systems
(MUPS) like avoiding dose dumping, tailored drug release profile, targeted to release at specific regions of the gastro-intestinal tract etc.
Capsules
a. Hard Gelatin Capsules
Powders, pellets, mini tablets or their combination can be filled into hard gelatin capsules
b. Soft Gelatin Capsules
Soft gelatin capsules can be developed for liquids or suspension filling for poorly soluble and thermolabile, photosensitive medicaments.
Powders for Suspension
We develop age appropriate, easily reconstitutable powders for oral suspensions which can be filled into sachets
Why Aurigene Solid Orals Services?
Services across the product lifecycle
US FDA inspected lab and manufacturing facilities
Experience with advanced formulation technologies
20+ years of experience in formulation
Connect with our scientific experts for your drug discovery, development, and manufacturing needs
We understand that clear communication is essential to successful collaborations, and that's why we have a dedicated team that is always ready to help you. Whether you have questions about our services, want to discuss a potential partnership, or simply want to learn more about our company, we're here to help.
Our team of experts is dedicated to providing personalised solutions tailored to your unique needs. So, please don't hesitate to reach out to us. We look forward to hearing from you and helping you achieve your business goals.

Methoxy Polyethylene Glycol (m-PEGs)
Aurigene Pharmaceutical Services is a leader in the synthesis of activated MethoxyPolyethyleneGlycol(m-PEGs), With a comprehensive product range and customized services. ...
Read MoreBase mediated spirocyclization of quinazoline: one-step synthesis of spiro-isoindolinone dihydroquinazolinones
2020
A novel approach for the spiro-isoindolinone dihydroquinazolinones has been demonstrated from 2- aminobenzamide and 2-cyanomethyl benzoate in the presence of KHMDS as a base to get moderate yields. The reaction has been screened in various bases followed by solvents and a gram scale reaction has also been executed under the given conditions. Based on the controll...
Read More-
Ultrasound assisted rapid synthesis of mefenamic acid based indole derivatives under ligand free Cu-catalysis: Their pharmacological evaluation
2005
An improved and rapid synthesis of mefenamic acid based indole derivatives has been achieved via the ligand free Cu-catalyzed coupling-cyclization method under ultrasound irradiation. This simple, straightforward and inexpensive one-pot method involved the reaction of a terminal alkyne derived from mefenamic acid with 2- iodosulfanilides in the presence of CuI ...
Read More -
Formal synthesis of Cladospolide C & epi-Cladospolide C using R- (þ)-g-valerolactone as a chiral synthon
2005
The formal synthesis of Cladospolide-C and its analog is achieved by using enantiopure (R)-g evalerolactone 10. The significant points of this synthesis are the stereoselective dihydroxylation of a, bunsaturated ester 16 using Sharpless protocol, Wittig olefination of g evalerolactol 6 with triphenylphosphonium iodide salt 7, one pot selective oxidation ...
Read More -
Amberlite-15 promoted an unprecedented aza Michael rearrangement for one pot synthesis of dihydroquinazolinone compounds
2005
A new one pot multicomponent annulation strategy for the synthesis of various dihydroquinazolinone compounds has been developed using Amberlite-15 as a catalyst, giving good to moderate yields. In this reaction the substrate scope for amines and aldehydes was also investigated. The reaction has been checked on a large scale ...
Read More
You are about to leave Aurigene Pharmaceutical Services and affiliates website. Aurigene Pharmaceutical Services assumes no responsibility for the information presented on the external website or any further links from such sites. These links are presented to you only as a convenience, and the inclusion of any link does not imply endorsement by Aurigene Pharmaceutical Services.
If you wish to continue to this external website, click Proceed.


Leaving already?
Don't forget to join us at
CPHI Worldwide 2023.
October 24th-26th, 2023 | Barcelona, Spain
Get ready to accelerate your drug’s journey to the market













